Top videos

Ellis demonstrates the need to drop the tip of the needle when withdrawing medication from a vial.
#NCLEX #NewGrad #ClinicalSkills #HESI #Kaplan #ATI #NursingSchool #NursingStudent #Nurse #RN #PN #Education #LPN #NurseEducator #lvn
🚨 Reminder: shipping deadlines are looming 👀
🎁 Regular Shipping: Order by Friday, December 15
🚀 Expedited Shipping: Order by Monday, December 18
🔍 Still searching for last-minute gifts? Consider a Level Up RN Gift Card! 💌 It’s not only a thoughtful present but also the perfect way to share treasures like Pharmacology Flashcards OR digital treasures like Flashables Digital Nursing Flashcards & the Level Up RN membership. Give the gift of knowledge this holiday season! 🧠⚡️💖 bit.ly/LevelUpRNGC
🚪 Access our Cram Courses, Quizzes and Videos all in one ad free space with Level Up RN Membership https://bit.ly/LevelUpRNMembership
Want more ways to MASTER Clinical Skills? Check out our flashcards & videos!
👇👇👇👇👇👇👇👇👇👇
👉 https://bit.ly/clinicalnursingskills 👈
☝️👆☝️👆☝️👆☝️👆☝️👆
This is your one-stop-shop for materials to help you LEARN & REVIEW so you can PASS Nursing School.
🤔🤔🤔 DO YOU WANT TO PASS your classes, proctored exams and the NCLEX? 🤔🤔🤔 Our resources are the best you can buy. They are built with a single goal: help you pass with no fluff. Everything you need, and nothing you don’t. Don’t take our word for it, though! Check out our hundreds of ⭐️⭐️⭐️⭐️⭐️ reviews from nurses who passed their exams and the NCLEX with Level Up RN.
🗂️ Our Ultimate Nursing School Survival kit is your number 1 resource to get through nursing school and to pass the NCLEX. Whether you're just starting school or you’re already prepping for the NCLEX, this bundle of flashcards is the best you can buy. It covers all the information you need to know to pass all your exams and it has FREE shipping!
➡️ https://bit.ly/TUNSSK ⬅️
L👀king for EVEN MORE resources to survive Nursing School? Make your Nursing School experience your own! Life’s difficult enough—learning shouldn’t be.
🪅 Games https://nursesquad.com
💻 Digital resources https://bit.ly/NursingStudyCourses
📅 Organizational tools https://bit.ly/OrganizingSchool
✨Want perks? Join our channel!
https://youtube.com/leveluprn/join
🏷 Head to https://leveluprn.com/specials for all our latest deals!🥳️
📧 LOOKING FOR FREE RESOURCES TO HELP WITH YOUR EXAMS? Get exclusive tips, latest video releases and more delivered to your email!
➡️ https://leveluprn.com/signup ⬅️
⚕ 👩 LEVEL UP NURSE SQUAD 👩⚕️
All of the nurses at Level Up RN are here to help! Cathy Parkes started helping her fellow classmates back when she was in nursing school, tutoring so they could pass their exams and graduate. After she got her BSN and started working as an RN at Scripps Encinitas Hospital, she started this YouTube channel to help nursing students around the world. Since then she has built a team of top-notch dedicated nurses and nurse educators who are focused on improving nursing education and supporting career advancement for nurses everywhere. With flashcards, videos, courses, organizational tools and more, we are singularly focused on helping students and nurses Level Up on their exams and nursing careers.
The epididymis is a long coiled tube that lies above and behind each testicle. The epididymis collects and transports sperm from the testis to the vas deferens (tubes that transport sperm to the urethra). An epididymal cyst is a cyst-like mass in the epididymis that contains clear fluid. Typically, epididymal cysts and spermatoceles do not cause symptoms. When discovered, the epididymal cyst is usually about the size of a pea and feels separate from the top of the testis. Spermatoceles typically arise from the head of the epididymis, and are felt on the top portion of the testicle. Epididymal cysts and spermatoceles are often incidental findings on testicular self-examination or routine physical examination. It is important that any mass noted in the scrotum be examined by a urologist in order to obtain an accurate diagnosis, especially a mass on the testicle itself. Our team in the Division of Urology will typically be able to confirm the diagnosis on physical exam. However, a scrotal ultrasound may also be used in order to rule out other conditions.
Breath sounds can be either normal or abnormal. These sounds come from the lungs when you breathe in or out. These sounds can be heard using a stethoscope or simply when breathing. Abnormal breath sounds can indicate a lung problem, such as: an obstruction inflammation an infection fluid in the lungs asthma Listening to breath sounds is an important part of diagnosing many different medical conditions.

Outpatient -- or same-day -- knee replacement surgery is more convenient than traditional knee replacement surgery and often can help you recover faster.
Outpatient -- or same-day -- knee replacement surgery is more convenient than traditional knee replacement surgery and often can help you recover faster. At Duke Ambulatory Surgery Center Arringdon, your knee replacement will be followed immediately by physical therapy to get you moving and start your recovery process right away. Our expert joint replacement team ensures your knee replacement surgery is safe and effective so you can return to the comfort of your home as soon as possible.

Supraventricular tachycardia (SVT) is an abnormal condition of heart which increases normal heartbeat rate rapidly. Normally, heartbeat rate should be between 80 to 100 beats per minute. For more info: https://goo.gl/14btbU

Obstructive lung diseases include conditions that make it hard to exhale all the air in the lungs. People with restrictive lung disease have difficulty fully expanding their lungs with air. Obstructive and restrictive lung disease share the same main symptom: shortness of breath with exertion.
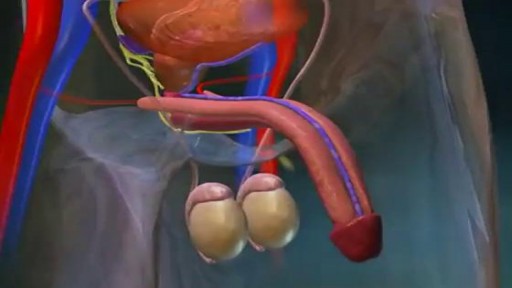
What Happens During an Erection?
In order to attain an erection, messages from the brain and other sense organs trigger the arteries of the penis to dilate. This allows an increased amount of blood to flow into three columns of spongy tissue in the penis.
As the arteries supplying blood to the corpus spongiosum and to the two larger columns, the corpus cavernosa, become filled with blood; the penis grows and becomes rigid. Pressure of the engorged tissue against the veins in the penis effectively traps blood within the penis until climax is reached or the sensation wanes.
What Are Penile Implants?
Impotence, or the inability to attain or maintain an erection, can be caused by a disruption at any stage in this process. Several types of penile implants are available that create an artificial erection. Two common types of implants are the semi-rigid malleable rod and the inflatable implant.
•The semirigid malleable rod is usually made of plastic with a core of flexible wire. These rods can be bent down to conceal the penis under clothing or raised to form an artificial erection.
•The inflatable implant is more complex and involves several working parts: a reservoir of fluid that is implanted into the abdomen, a pump system located in the scrotal sac near the testes, and two inflatable cylinders.
How Penile Implants Help Erectile Fuctioning
In order to attain an erection, the scrotal pump must be squeezed repeatedly to propel fluid into the penile cylinders. When an erection is no longer desired, a release valve is pressed on the side of the pump and the cylinders deflate.
Before Having Penile Implant Surgery
Persons considering these types of implants should speak with their physician or healthcare professional about possible risks and complications.

Anytime you're having unprotected sex, there's always a chance that a woman can get pregnant. Pregnancy requires sperm and egg to meet up together so a woman needs to be during her most fertile time of the month, which is usually 6 days out of the month; 5 days leading up to ovulation and on the day of ovulation. For most women, ovulation happens 12-16 days before her period's going to start. So a woman is usually most fertile for a week to a week and a half after her period has ended generally speaking, if you don't want to count each and every single day. So if you have unprotected intercourse during this time, then there's a high probability that a woman can get pregnant. Now, you mentioned that your girlfriend is supposed to start her period in about five days or so. If you've had intercourse any time leading up to this, there's always a chance that she could get pregnant. But as for the mechanics of it all, in order to get pregnant, semen needs to be inserted inside the vaginal canal where the egg and sperm can then meet. So if that did not happen, then the chances of her getting pregnant are slim. But if that has happened, the chances of her getting pregnant are great. So it would be best for you and her to just wait until her period is supposed to start and if she's late, then take an over-the-counter pregnancy test and if it's positive, congratulations to both of you! If it's negative and she still doesn't start her period, then tell her to wait about 5-7 days. Take another test and then maybe at that point, it will be positive if she is indeed pregnant. If she continues to not have a period or she's concerned about anything, it would be best for her to follow up with her doctor and they can decide if further investigation or treatment is warranted. If you have any other questions for me, feel free to ask them on our Facebook page at facebook.com/intermountainmoms and recommend us to your friends and family, too.

Adult Still's disease is a rare type of inflammatory arthritis that features fevers, rash and joint pain. Some people have just one episode of adult Still's disease. In other people, the condition persists or recurs. This inflammation can destroy affected joints, particularly the wrists. Treatment involves medications, such as prednisone, that help control inflammation