Top videos
Stapling is used to treat prolapsed hemorrhoids. A surgical staple fixes the prolapsed hemorrhoid back into place inside your rectum and cuts off the blood supply so that the tissue will shrink and be reabsorbed. Stapling recovery takes less time and is less painful than recovery from a hemorrhoidectomy.

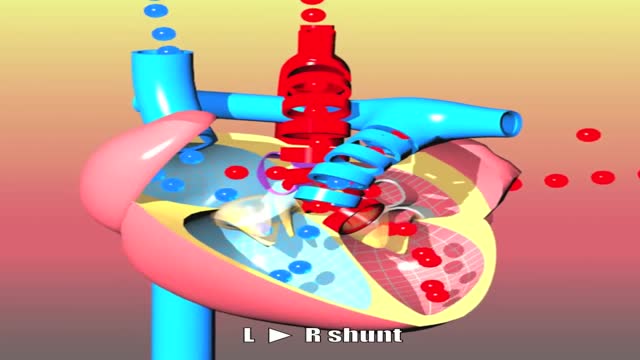
It is a specialized bodily fluid that supplies essential substances and nutrients, such as sugar, oxygen, and hormones to our cells, and carries waste away from those cells, this waste is eventually flushed out of the body in urine, feces, sweat, and lungs (carbon dioxide). Blood also contains clotting agents.
Recovery can take 4 to 6 months, depending on the size of the tear and other factors. You may have to wear a sling for 4 to 6 weeks after surgery. Pain is usually managed with medicines. Physical therapy can help you regain the motion and strength of your shoulder.

While it is unclear whether high heel shoes may or may not cause back pain, it is common for high heels to exacerbate an already present spinal condition. ... This pain in the back may also result from foot or leg fatigue that results from wearing these shoes and this can affect whole body mechanics.
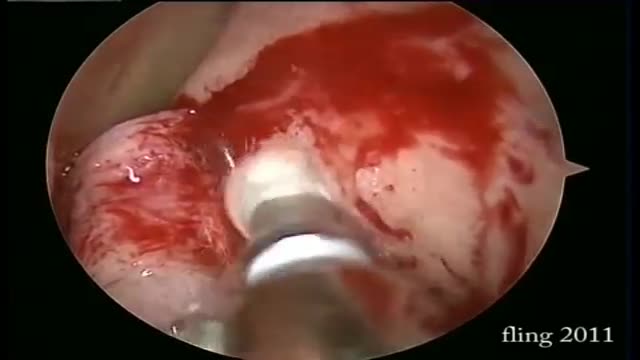
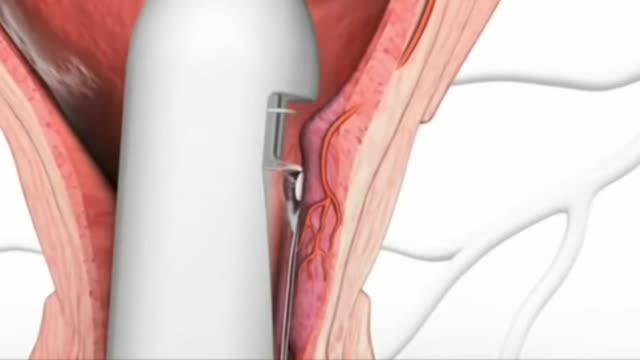
Dacryocystorhinostomy (DCR) is a procedure performed for the treatment of tearing (epiphora) due to blockage of the nasolacrimal duct. Tears originate in the lacrimal gland, located at the upper outer margin of the eye. As tears cross the eye with each blink, they are directed into small openings in the eyelids called puncta. From this point, tears travel through a pathway known as the canalicular system into the lacrimal sac. The lacrimal sac is located between the eye and the nose, and funnels tears into the nasal cavity through the nasolacrimal duct (Figure 1). As this is quite a long path for tears to travel, there can be many causes of excessive tearing. Blockage of the nasolacrimal duct is one common cause, and can be treated by creating a direct opening from the lacrimal sac into the nasal cavity in a procedure known as DCR. The evaluation and management of tearing may involve both an ophthalmologist and an otolaryngologist.

Marfan syndrome is a genetic disorder that affects the body’s connective tissue. Connective tissue holds all the body’s cells, organs and tissue together. It also plays an important role in helping the body grow and develop properly. marfan_general_2.jpg What is Marfan Syndrome?Connective tissue is made up of proteins. The protein that plays a role in Marfan syndrome is called fibrillin-1. Marfan syndrome is caused by a defect (or mutation) in the gene that tells the body how to make fibrillin-1. This mutation results in an increase in a protein called transforming growth factor beta, or TGF-β. The increase in TGF-β causes problems in connective tissues throughout the body, which in turn creates the features and medical problems associated with Marfan syndrome and some related disorders. Because connective tissue is found throughout the body, Marfan syndrome can affect many different parts of the body, as well. Features of the disorder are most often found in the heart, blood vessels, bones, joints, and eyes. Some Marfan features – for example, aortic enlargement (expansion of the main blood vessel that carries blood away from the heart to the rest of the body) – can be life-threatening. The lungs, skin and nervous system may also be affected. Marfan syndrome does not affect intelligence.

A visual prosthesis, often referred to as a bionic eye, is an experimental visual device intended to restore functional vision in those suffering from partial or total blindness. In 1983 Joao Lobo Antunes, a Portuguese doctor, implanted a bionic eye in a person born blind.