Top videos
This surgery is usually done while you are under general anesthesia. That means you will be asleep and pain-free. Healthy skin is taken from a place on your body called the donor site. Most people who are having a skin graft have a split-thickness skin graft. This takes the two top layers of skin from the donor site (the epidermis) and the layer under the epidermis (the dermis). The donor site can be any area of the body. Most times, it is an area that is hidden by clothes, such as the buttock or inner thigh. The graft is carefully spread on the bare area where it is being transplanted. It is held in place either by gentle pressure from a well-padded dressing that covers it, or by staples or a few small stitches. The donor-site area is covered with a sterile dressing for 3 to 5 days. People with deeper tissue loss may need a full-thickness skin graft. This requires an entire thickness of skin from the donor site, not just the top two layers. A full-thickness skin graft is a more complicated procedure. Common donor sites for full-thickness skin grafts include the chest wall, back, or abdominal wall.
Indications for intervention in patients with a renal artery aneurysm (RAA) include the following [20, 8, 13, 14] : Rupture Symptomatic RAA - Hypertension (from associated renal artery stenosis, refractory to medical management), pain, renal ischemia or infarction secondary to embolization from the aneurysm sac RAAs in females who are pregnant or are contemplating pregnancy Diameter greater than 2 cm Enlarging RAA RAA associated with acute dissection Currently, there is no consensus regarding the size at which an RAA should be repaired in an asymptomatic patient. Experts have recommended RAA repair at diameters ranging from 1.5 to 3 cm, [8] though most suggest 2 cm. Some reports have even suggest that larger asymptomatic saccular aneurysms may be managed expectantly. Note that aneurysm rupture at a diameter of 1.5 cm has been reported. Complete calcification of the wall of the aneurysm sac manifests in about 40% of patients. This was once believed to confer protection against rupture [21] ; however, this belief has since been questioned. [30] Asymptomatic, small (<2 cm in diameter) RAAs do not usually require treatment. One notable exception is an RAA in a woman who is pregnant or contemplating pregnancy. In view of the increased risk of rupture in such cases, even small asymptomatic aneurysms should be repaired in this population. For diagnosis and preinterventional planning, gadolinium-enhanced magnetic resonance angiography (MRA) and computed tomography (CT) angiography (CTA) with three-dimensional (3D) reconstruction have essentially replaced conventional arteriography. Regular follow-up examination with ultrasonography (US) or CT) is recommended in patients who are treated expectantly. Spontaneous cure by thrombosis of small aneurysms has been described. Further refinements in endovascular techniques may allow more RAAs to be treated in this manner. So far, excellent short- and intermediate-term results have been described in the literature [40] ; however, there remains a need for further long-term outcome data.

The best way to prevent hepatitis A is through vaccination with the hepatitis A vaccine. Vaccination is recommended for all children age 12 months or older, for travelers to certain countries, and for people at high risk for infection with the virus. The hepatitis A vaccine is given as two shots, six months apart.
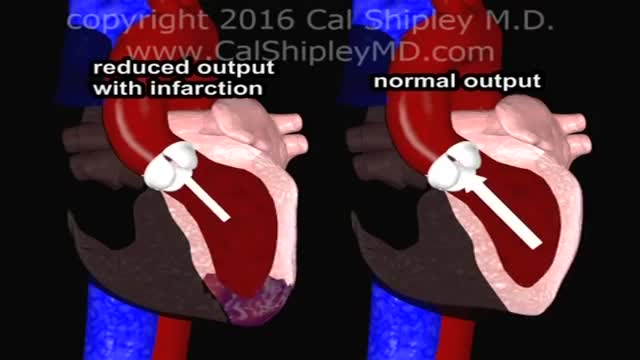
Cardiogenic shock is a condition in which your heart suddenly can't pump enough blood to meet your body's needs. The condition is most often caused by a severe heart attack. Cardiogenic shock is rare, but it's often fatal if not treated immediately. If treated immediately, about half the people who develop the condition survive.
Surgery to repair a torn rotator cuff most often involves re-attaching the tendon to the head of humerus (upper arm bone). A partial tear, however, may need only a trimming or smoothing procedure called a debridement. A complete tear is repaired by stitching the tendon back to its original site on the humerus.
This short course reviews the main features of EKG tracings. A method for analyzing EKGs is also presented. This method includes assessment of rhythm, calculating heart rate, observing P-wave forms, measurement of EKG intervals and segments and the evaluation of other relevant waves.
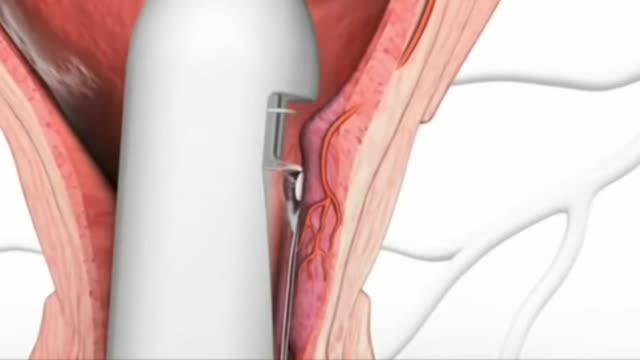
Stapling is used to treat prolapsed hemorrhoids. A surgical staple fixes the prolapsed hemorrhoid back into place inside your rectum and cuts off the blood supply so that the tissue will shrink and be reabsorbed. Stapling recovery takes less time and is less painful than recovery from a hemorrhoidectomy.

It is a specialized bodily fluid that supplies essential substances and nutrients, such as sugar, oxygen, and hormones to our cells, and carries waste away from those cells, this waste is eventually flushed out of the body in urine, feces, sweat, and lungs (carbon dioxide). Blood also contains clotting agents.
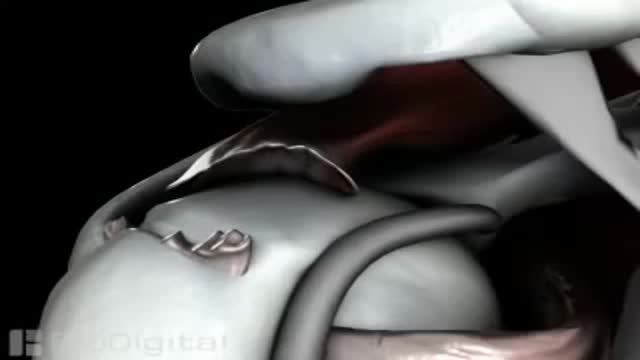
Recovery can take 4 to 6 months, depending on the size of the tear and other factors. You may have to wear a sling for 4 to 6 weeks after surgery. Pain is usually managed with medicines. Physical therapy can help you regain the motion and strength of your shoulder.

Marfan syndrome is a genetic disorder that affects the body’s connective tissue. Connective tissue holds all the body’s cells, organs and tissue together. It also plays an important role in helping the body grow and develop properly. marfan_general_2.jpg What is Marfan Syndrome?Connective tissue is made up of proteins. The protein that plays a role in Marfan syndrome is called fibrillin-1. Marfan syndrome is caused by a defect (or mutation) in the gene that tells the body how to make fibrillin-1. This mutation results in an increase in a protein called transforming growth factor beta, or TGF-β. The increase in TGF-β causes problems in connective tissues throughout the body, which in turn creates the features and medical problems associated with Marfan syndrome and some related disorders. Because connective tissue is found throughout the body, Marfan syndrome can affect many different parts of the body, as well. Features of the disorder are most often found in the heart, blood vessels, bones, joints, and eyes. Some Marfan features – for example, aortic enlargement (expansion of the main blood vessel that carries blood away from the heart to the rest of the body) – can be life-threatening. The lungs, skin and nervous system may also be affected. Marfan syndrome does not affect intelligence.

A visual prosthesis, often referred to as a bionic eye, is an experimental visual device intended to restore functional vision in those suffering from partial or total blindness. In 1983 Joao Lobo Antunes, a Portuguese doctor, implanted a bionic eye in a person born blind.
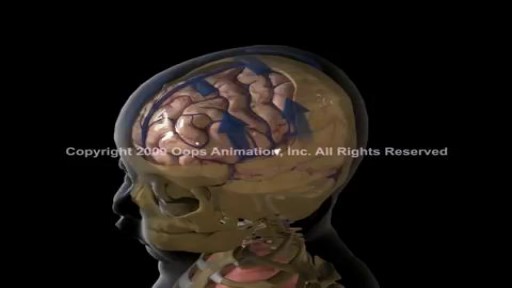
Shaken baby syndrome usually occurs when a parent or caregiver severely shakes a child in frustration or anger, often because the child won't stop crying. Permanent brain damage or death may result. Symptoms include irritability, difficulty staying awake, seizures, abnormal breathing, poor eating, bruises, and vomiting. Treatment will vary depending on complications related to shaken baby syndrome. In rare instances, breathing support or surgery may be needed to stop bleeding in the brain.