Top videos

How to Use Wash your hands. Check the drug label to be sure it is what your doctor prescribed. ... Remove pen cap. Look at the insulin. Wipe the tip of the pen where the needle will attach with an alcohol swab or a cotton ball moistened with alcohol.

Fulminant hepatic failure (FHF) or acute liver failure (ALF) is defined as the rapid development of acute liver injury with severe impairment of the synthetic function and hepatic encephalopathy in a patient without obvious, previous liver disease.

Function. Vitamin A helps form and maintain healthy skin, teeth, skeletal and soft tissue, mucus membranes, and skin. It is also known as retinol because it produces the pigments in the retina of the eye. Vitamin A promotes good vision, especially in low light. Vitamin deficiency anemia occurs when your body doesn't have enough of the vitamins needed to produce adequate numbers of healthy red blood cells. Red blood cells carry oxygen from your lungs throughout your body. If your diet is lacking in certain vitamins, vitamin deficiency anemia can develop.

The foods for your child are easily digestible foods, such as rice cereal, pasta, breads, cooked beans, mashed potatoes, cooked carrots, applesauce, and bananas. Pretzels or salty crackers can help your child replace the salt lost from diarrhea. Foods containing large amounts of sugar or fat should be avoided.
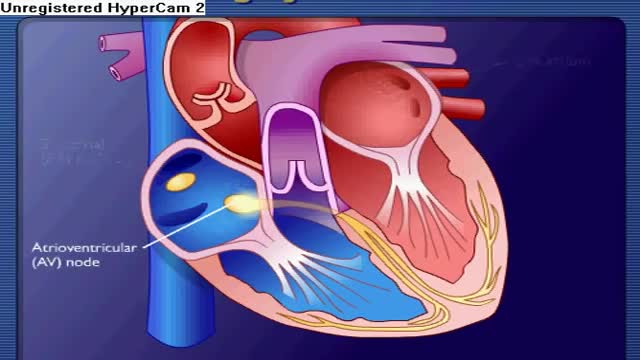
Premature ventricular contractions (PVCs) are extra, abnormal heartbeats that begin in one of your heart's two lower pumping chambers (ventricles). These extra beats disrupt your regular heart rhythm, sometimes causing you to feel a flip-flop or skipped beat in your chest.
A new report analyzing FDA-approved monoclonal antibodies (mAbs) produced by a select group of leading biotechnology companies shows that clinical development times – specifically the duration of Phase II and Phase III trials – are lengthening, while FDA review times have remained constant. The average time from investigational new drug (IND) filing to market was 6.7 years for 11 mABs approved between 1994 and 2003 but shot up to 8.3 years for 12 mAbs approved between 2004 and March 9, 2011, according to Deloitte Recap LLC’s analysis, Therapeutic Monoclonal Antibodies – Insights, Strategies and Data.
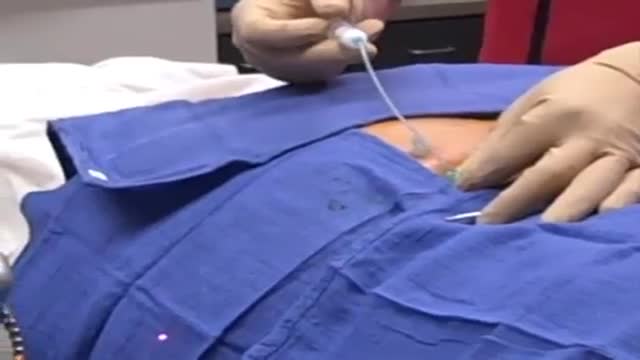
A facet joint injection is a relatively simple, straightforward procedure, and is usually performed in an office based procedure suite or in an ambulatory surgical center. As with many spinal injections, facet joint injections are best performed using fluoroscopy (live X-ray) for guidance to properly target and place the needle (and to help avoid nerve injury or other injury).

Rheumatic fever is an inflammatory disease that can develop as a complication of inadequately treated strep throat or scarlet fever. Strep throat and scarlet fever are caused by an infection with streptococcus bacteria. Rheumatic fever is most common in 5- to 15-year-old children, though it can develop in younger children and adults. Although strep throat is common, rheumatic fever is rare in the United States and other developed countries. However, rheumatic fever remains common in many developing nations. Rheumatic fever can cause permanent damage to the heart, including damaged heart valves and heart failure. Treatments can reduce damage from inflammation, lessen pain and other symptoms, and prevent the recurrence of rheumatic fever.
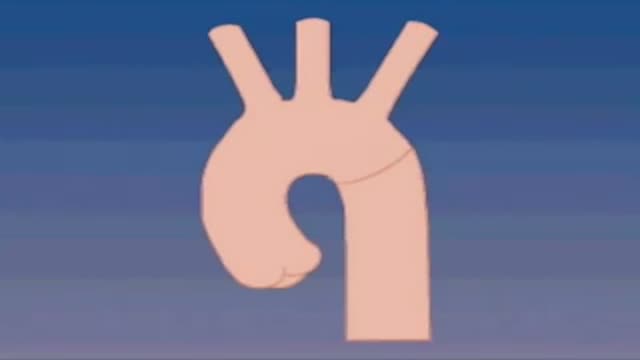
A narrowing of the major artery (the aorta) that carries blood to the body. This narrowing affects blood flow where the arteries branch out to carry blood along separate vessels to the upper and lower parts of the body. CoA can cause high blood pressure or heart damage.
Ehlers-Danlos syndrome is a group of disorders that affect the connective tissues that support the skin, bones, blood vessels, and many other organs and tissues. Defects in connective tissues cause the signs and symptoms of Ehlers-Danlos syndrome, which vary from mildly loose joints to life-threatening complications. Previously, there were more than 10 recognized types of Ehlers-Danlos syndrome, differentiated by Roman numerals. In 1997, researchers proposed a simpler classification that reduced the number of major types to six and gave them descriptive names: the classical type (formerly types I and II), the hypermobility type (formerly type III), the vascular type (formerly type IV), the kyphoscoliosis type (formerly type VIA), the arthrochalasia type (formerly types VIIA and VIIB), and the dermatosparaxis type (formerly type VIIC). This six-type classification, known as the Villefranche nomenclature, is still commonly used. The types are distinguished by their signs and symptoms, their underlying genetic causes, and their patterns of inheritance. Since 1997, several additional forms of the condition have been described. These additional forms appear to be rare, affecting a small number of families, and most have not been well characterized.

The DASH diet is a lifelong approach to healthy eating that's designed to help treat or prevent high blood pressure (hypertension). The DASH diet encourages you to reduce the sodium in your diet and eat a variety of foods rich in nutrients that help lower blood pressure, such as potassium, calcium and magnesium.

The menstrual cycle is the regular natural change that occurs in the female reproductive system like the uterus and ovaries that make pregnancy possible. The cycle is required for the production of ovocytes, and for the preparation of the uterus for pregnancy.

The syndrome of inappropriate antidiuretic hormone (ADH) secretion (SIADH) is defined by the hyponatremia and hypo-osmolality resulting from inappropriate, continued secretion or action of the hormone despite normal or increased plasma volume, which results in impaired water excretion.

Adrenoleukodystrophy, or ALD, is a deadly genetic disease that affects 1 in 18 000 people. It most severely affects boys and men. This brain disorder destroys myelin, the protective sheath that surrounds the brain's neurons -- the nerve cells that allow us to think and to control our muscles.